What Is Simple Diffusion?
In an answer or fuel online that has a place of excessive numbers of debris and a place of decrease numbers of debris, the debris will diffuse, or flow, from the location of better to the location of decrease awareness. This is thought as easy diffusion, or passive diffusion.
Simple diffusion does now no longer require electricity. Instead, it takes place via way of means of random movement. Random movement is described as motion that takes place via way of means of hazard due to the fact there’s no order or everyday gadget via way of means of which the actions of the debris shift in an answer or fuel online.
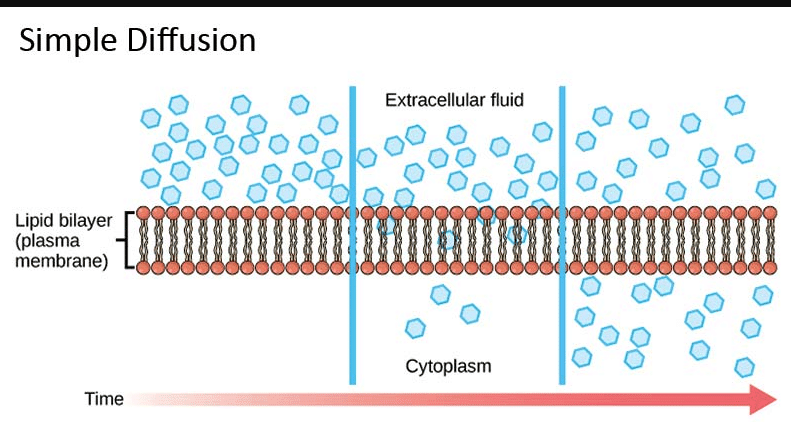
These regions of better or decrease numbers of debris in an answer or fuel online are separated via way of means of a membrane. A membrane is a skinny substance that can permit materials to by skip via it or now no longer. Therefore, a membrane is assessed as permeable (because of this that matters can by skip via it), impermeable (because of this that matters can not by skip via it), or semi permeable (because of this that matters can by skip via it via way of means of diffusion). In the case of easy diffusion, the membrane is semi permeable, because of this that the debris or solutes can by skip via it via way of means of random movement.
More details
To assist you apprehend this system, think about a small brook with flowing water. Imagine that the brook has small leaves and different particles within side the water. Then consider there’s a window display within side the brook. The display represents the semi permeable membrane, and the leaves and different particles within side the water constitute debris. In this case, small leaves and particles can by skip via the display at the same time as large leaves and particles can not by skip.
Read Also: What are Capital Resources?
So, let’s positioned all of it together. Concentration gradient is the system via way of means of which debris, which might be now and again referred to as solutes, flow via an answer or fuel online from a place of better quantity of debris to a place of decrease quantity of debris. The real motion is refer to as diffusion. Solutes transferring via an answer or a fuel online take place via way of means of random movement alongside the awareness gradient till there are same numbers of debris within side the regions.
Diffusion of electrolytes
- The precept for the diffusion of charged species is pushes via way of means of an extra pressure together with the awareness gradient.
- Charged solutes are situation to electric forces whilst electrostatic capability gradients are present.
- Accordingly, the using pressure for electrolyte delivery is the gradient of the electrochemical capability in place of that of the chemical capability.
- Since any electrolyte answer should incorporate at the least one anion and one cation, there are continually at the least solute species which ends up in a couple of fluxes.
Factors affecting Simple diffusion
Because the charge of diffusion is decided via way of means of numerous parameters, those parameters/elements have an effect on the mechanism of diffusion.
-
Concentration gradient
- The awareness gradient throughout a organic membrane is the using pressure for the diffusion of a nonelectrolyte.
- Therefore, the better the awareness distinction throughout the membrane, the better may be the charge of diffusion.
- As the distribution of molecules throughout the membrane movements closer to uniformity, the charge of diffusion decreases.
- Once equilibrium is maintain throughout the membrane, the system of diffusion ceases.
-
Mass/Size of the solute molecules
- The length of the molecules additionally influences the charge of diffusion throughout a organic membrane.
- If the dimensions of the molecules is large, it will likely be greater hard for it to transport throughout the membrane, which, in turn, slows the charge of diffusion of the molecule.
- Thus, the charge of diffusion is better for smaller molecules and slower for large molecules.
-
Temperature
- The temperature of the gadget additionally influences the system of easy diffusion.
- With the boom in temperature, the electricity of the molecules additionally will increase.
- Molecules with better electricity can flow quicker throughout the membrane at the same time as debris with decrease electricity flow slower.
-
Solubility
- The solubility of molecules in a medium additionally influences the charge of diffusion of the debris.
- Molecules which might be lipid-soluble can flow quick throughout the lipid layer just like the plasma membrane.
- Similarly, polar and non-polar molecules additionally flow with a extraordinary charge relying on the character of the organic membrane.
-
Solvent density
- With the boom in solvent density, the charge of diffusion decreases.
- More dense a solvent, greater hard it will likely be for the solute to transport around.
- Solvent density performs an critical position withinside the motion of solute withinside the cytoplasm of the cell.
- An boom within side the density of the cytoplasm slows down the motion of the molecules and gases, and the opposite is authentic for much less dense cytoplasm.
-
Surface location and thickness of the organic membrane
- The charge of diffusion boom with the boom within side the floor location of the membrane.
- The boom in floor location will increase the permeability or mobility of the molecules. As mobility is one of the elements accountable for the flux.
- Similarly, the charge of diffusion is likewise decreases. Via way of means of the growing thickness of the membrane.
Simple Diffusion Process
There are in reality 3 styles of diffusion. We’ve been speak me approximately easy diffusion. Additionally refer to as passive diffusion. However there may be additionally facilitative diffusion and lively transport. We will most effective move into element approximately the focal point of this lesson: easy diffusion.


